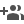A hybrid electrode-based flexible symmetric supercapacitor that shows excellent electrochemical properties, cycle stability and high energy density could bring a future solution for efficient energy storage.
Efficient and stable electrode materials for energy storage have always been a hot topic of research as the energy requirements grow day-by-day. The primary goal of researchers working in supercapacitor electrodes is to push the energy density values close to those of batteries. Conducting polymers are pseudocapacitive materials with multi-faceted applications and are capable of delivering the basic requirements for supercapacitor electrodes.
The Materials for Energy Storage and Optoelectronic Devices Group in the Department of Physics, Sanatana Dharma College, Alappuzha has developed a hybrid electrode-based flexible symmetric supercapacitor that shows excellent electrochemical properties, cycle stability and high energy density. The binder-free hybrid electrode consists of high-molecular weight polyaniline (PANi) prepared via self-stabilized dispersion polymerization and vanadium pentoxide nanostructures prepared by a facile microwave assisted method.
Unlike the conventional PANi based electrodes prepared using the emeraldine base powder cast on flexible substrates as slurry, here the electrodes are prepared from a dispersion of PANi with a secondary dopant in m-cresol. One of the reasons for the excellent performance of the hybrid electrode is this unique preparation method that combines flexible and conducting, high-molecular weight PANi prepared by an organic solvent mediated self-stabilized polymerisation method and highly stable V2O5 nanostructures showing excellent rate-capability making it usable in a wide range of input current values.
The incorporation of this high molecular weight PANi with nanostructured V2O5 effectively addresses the shortcomings of these individual materials and exhibits a synergistic effect as evident from the performance of the hybrid electrode. The flexible supercapacitor device made using these electrodes exhibit superior electrochemical characteristics with very high energy density and cycling stability, being the highest among the values reported for supercapacitors using aqueous electrolytes.
This research work carried out at SD College using the instrumentation facility provided by the Department of Science and Technology (DST) through the Fund for Improvement of S&T Infrastructure (FIST) programme, was recently published in the Chemical Engineering Journal.
Link to the article: https://doi.org/10.1016/j.cej.2023.143499