A new composite electrode material has been developed that can prevent cracks and damages in aluminium ion batteries and make them affordable, safe, and long-lasting.
As the world searches for safer and more eco-friendly alternatives to lithium-ion batteries, aluminium-based batteries are gaining increasing attention. Aluminium is one of the most common metals on Earth. It is easy to find, inexpensive, and it can store a large amount of electrical charge per atom than lithium. Because of these advantages, aluminium batteries could become an attractive option for future energy storage technologies. However, developing practical aluminium batteries has been challenging. The material inside these batteries breaks down quickly during charging and discharging. Over time, the material that stores energy can crack or dissolve into the liquid inside the battery causing the battery to lose its power fast.
A popular material researchers used for cathode is vanadium oxide. This material can store a large amount of energy and its layered structure allows aluminium ions to move in and out during battery operation. But, in water based aluminium batteries, this material tends to dissolve into the battery liquid. As a result, the battery loses capacity very quickly and cannot maintain stable performance for long periods.
To address this long-standing challenge, a research team led by Dr. Kavita Pandey from the Centre for Nano and Soft Matter Sciences (CeNS), Bengaluru, an autonomous institute of the Department of Science and Technology (DST), Government of India, worked in collaboration with researchers from Shiv Nadar Institution of Eminence, Greater Noida.
The team combined vanadium oxide with another layered material known as MXene. MXene is an extremely thin material that can conduct electricity very efficiently. When these two materials are combined, the MXene forms a supportive and conductive network that helps hold the vanadium oxide in place. This structure also creates smooth pathways that allow aluminium ions to move more easily during battery operation.
This design helps reduces the amount of vanadium that dissolves into the battery liquid. Quantitative analysis revealed that vanadium dissolution in the composite material was suppressed to just 5.4 ppm, compared to 28.3 ppm for pristine V2O5, direct evidence of the MXene scaffold's stabilizing role. Because of this improved stability, the battery was able to operate for a much longer time. Electrochemical tests showed that the battery maintained more than 73% of its original capacity after 100 charging cycles and even after 500 cycles , it still held at about 59%. In comparison, batteries made using the original material lose their energy storage ability much more quickly.
The team studied the battery material using XRD, XPS and electron microscopy to understand why it works well even after repeated use. These studies revealed that the MXene framework helps maintain the structure of the electrode, preventing cracks and damages normally occurring during the battery operation.
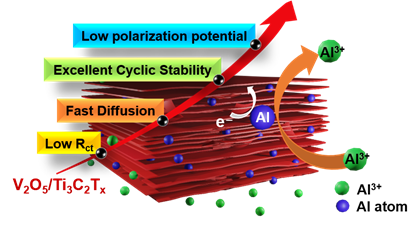
Fig: Schematic representation of the V2O5/MXene nanocomposite cathode for an aqueous aluminium-ion battery, where the conductive scaffold enables enhanced Al3+ kinetics.
Further, computer simulation studies showed that the MXene layer does much more than simply help electricity flow through the material. When the two materials come together, their interaction improves the movement of electrical charges and creates conditions that allow aluminium ions to move more easily and settle into stable positions within the structure. The simulations further revealed that this interaction changes how the material stores aluminium ions. The MXene layers help create an internal electric environment that speeds up the movement of charge inside the battery and makes certain oxygen sites within the material particularly suitable for holding aluminium ions. This helps the structure remain stable and prevents the active material from dissolving into the battery liquid.
These research findings offer an important guideline for designing stronger and longer-lasting materials for future aluminium batteries that are affordable, safe, and long-lasting.
The findings of this research have been published in the Journal of Power Sources.
Publication link: https://doi.org/10.1016/j.jpowsour.2026.239635
For further information, please contact: Dr. Kavita Pandey Email: kavitapandey[at]cens[dot]res[dot]in